The most common one is water vapour, H 2O. Most gases, however, are compounds with two or more different elements chemically united. A monatomic gas is one in which the basic unit is a single atom. They are summarized in the chart below.Ī diatomic gas is one in which the basic unit is a molecule made of two atoms joined together. Solids and liquids are more obviously matter: We can see that they. Matter is defined as anything that occupies space and has mass, and it is all around us. To know that in all the states of aggregation the matter has mass and takes a place although depending on the state, can have or not, form and volume fixed. Define and give examples of atoms and molecules. The symbols of the elemental gases can be found in any Periodic Table of Elements. Classify matter as an element, compound, homogeneous mixture, or heterogeneous mixture with regard to its physical state and composition. There are elemental gases (made up of a single element) and gases that are compounds (made up of more than one element). 
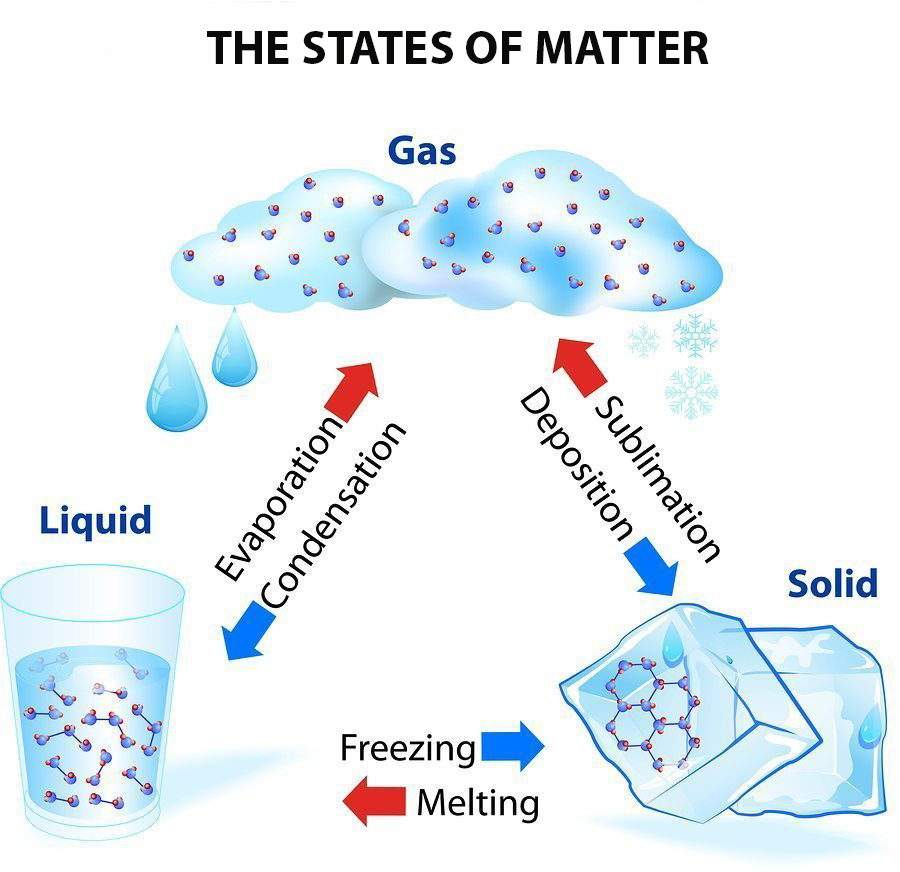
1: Matter is usually classified into three classical states, with plasma sometimes added as a fourth state. Representation of a gas (©2020 Let’s Talk Science). Matter typically exists in one of three states: solid, liquid, or gas. 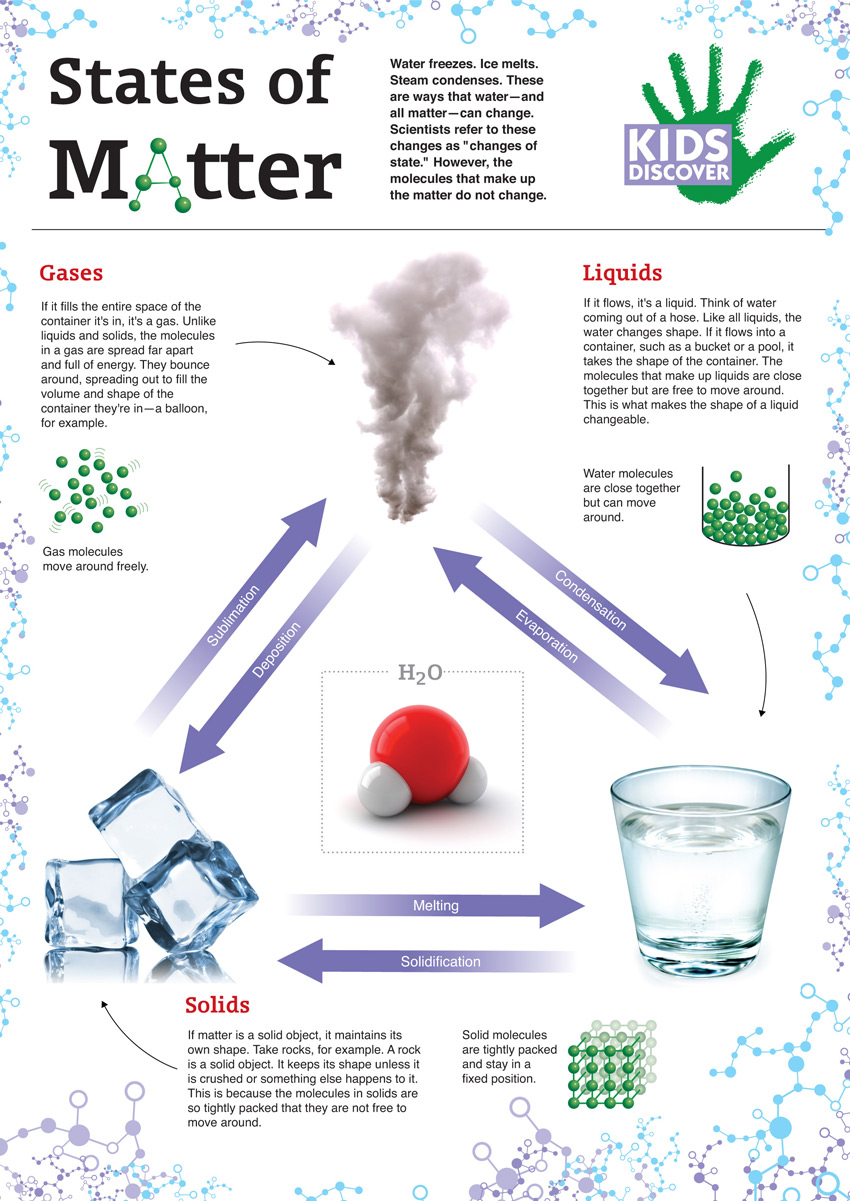
Open Professional Learning × Close Professional Learning It is the only metal we know of that is liquid at room temperature.Open Educational Resources × Close Educational Resources On earth, matter commonly exists in three states: solids, of fixed shape and volume. They vibrate in place but don’t move around. In a solid, the atoms and molecules are attached to each other. There are three common states of matter: Solids relatively rigid, definite volume and shape. In many substances, atoms are combined into molecules. Figure 6 (Left) A representative position space quasiparticle wave function (the 12th excited single quasiparticle state) for the symmetric SIAM with 0. A state of matter is a way to describe the behaviour of atoms and molecules in a substance. The basic building block of matter is the atom, the smallest unit of an element that can enter into combinations with atoms of the same or other elements. Particles are free to move over each other, but are still attracted to each otherĪ familiar liquid is mercury metal. Matter is anything that occupies space and has mass.No definite shape (takes the shape of its container).Liquids have the following characteristics: However, because the particles can move about each other rather freely, a liquid has no definite shape and takes a shape dictated by its container. The most obvious physical properties of a. In a liquid, the particles are still in close contact, so liquids have a definite volume. Liquid, in physics, one of the three principal states of matter, intermediate between gas and crystalline solid. If the particles of a substance have enough energy to partially overcome intermolecular interactions, then the particles can move about each other while remaining in contact. Images used with permission (public domain) Note that, as in the crystal, each Silicon atom is bonded to 4 oxygen atoms, where the fourth oxygen atom is obscured from view in this plane. The state that a given substance exhibits is also a physical property. From left to right: quartz (solid), water (liquid), nitrogen dioxide (gas). 
(right) The random network structure of glassy \(SiO_2\) in two-dimensions. Matter typically exists in one of three states: solid, liquid, or gas. \): (left) The periodic crystalline lattice structure of quartz \(SiO_2\) in two-dimensions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
